Scientists and engineers at the molecular spectroscopy laboratory conduct research in several scientific fields.
1). A study of the molecular dynamics of plant polymers using cotton labels as an example of spin labels.
(Ph.D., Associate Professor I.Kh. Yusupov, Ph.D., Senior Researcher A.D. Bakhdavlatov).
Brief information about the work performed.
The spin label method was used to study the effect of humidity on the molecular dynamics of white cotton fiber of the Astrakhan-2013 variety obtained from seeds modified with a biologically active solution. It is shown that the spectral and dynamic parameters characterizing the mobility of the spin label depend on the degree of moisture of the samples. It was found that at a relative humidity of P / Ps ≥ 0.60 new components of the EPR spectrum (h ’and h’ ’’) appear, which belong to the rapidly rotating radical, which indicates the appearance of structural defects and structural transitions in the cotton fiber.
The physicomechanical properties of polymers are largely dependent on the dynamics of macromolecular chains.
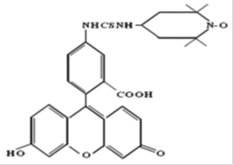
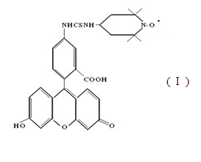
An effective method for studying the conformational mobility of macromolecules is the spin label method. It was found that the mobility parameters of spin-nitroxyl labels introduced into the polymer structure reflect the corresponding dynamics of the polymer matrix in the region of their attachment. The conformational mobility of macromolecular chains in polymer fibers of medium-fiber cotton fiber of the Astrakhan-2 (A-2) selection variety obtained from seeds modified with a biologically active solution (patented by VNIIOB RF) in an encapsulated system depending on the relative moisture content P / Ps was studied . The samples studied were reps of the VNIIOB Astrakhan RF employees. The fibers were chemically modified by hydroxyl groups (OH) with a spin label. The first derivative of the EPR absorption signal V1 was recorded on an EPR-1306 spectrometer. The relative humidity of the samples (P / Ps: 0.04; 0.060; 0.96) was set by placing them for five days in a special chamber that communicated with a solution of sulfuric acid of a certain density. A stable nitroxyl radical (I) having the following structural
the formula:
Typical experimental spectra are shown in Fig. 1.
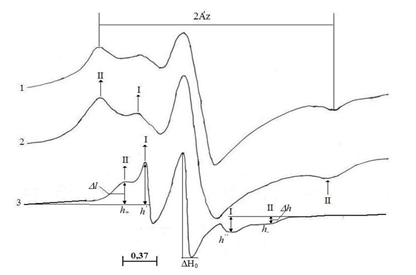
Fig. 1. EPR – spectra of spin-labeled cotton fiber depending on the content of relative humidity P / Ps: 1 – 0.04; 2-0.60; 3-0.96 at room temperature.
The line shape parameters of standard spectra of spin-labeled fibers are sensitive to sample moisture. In fig. Figures 2 and 3 show the dependences of the parameters Δl (half-width of the line in a low field) 2A’z (distances between external extrema), h ‘/ h (the ratio of the amplitudes of the low field lines of weakly and strongly immobilized labels) and ΔH0 (the width of the central component) on relative humidity P / Ps. In the case of dry samples (P / Ps = 0.04), a monotonic change in the parameters h ’/ h, 2A’z and ∆l (Figs. 2 and 3) is observed, indicating the amplification of low-amplitude high-frequency vibrations of the radical. The change in the parameters 2A’z (see Fig. 2, curve 2) and ΔH0 (see Fig. 3, curve 2) in the interval P / Ps ≤ 0.5 is the same slightly, and when the parameter h’/h increases, at the same time A sharp decrease in the parameters 2A’z and ΔH0 is observed in the region P / Ps = 0.5 – 0.6 (Figs. 2 and 3).
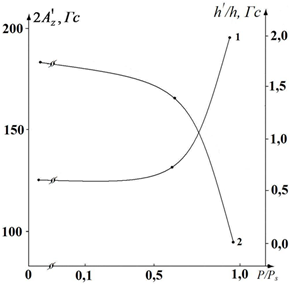
Fig. 2. Dependences of the parameters of the EPR spectrum and h ’/ h on the relative humidity P / Ps of cotton fiber samples, grade A-2. From the graph of the dependence of the EPR spectrum parameter h ‘/ h on humidity, it can be seen that for P / Ps ≥ 0.5 the value of this parameter increases (Fig. 2 curve 1), which indicates an increase in the label mobility in defects in the fiber structure with τ0 ≤ 10 -8 s – 5 • 10 -7s.
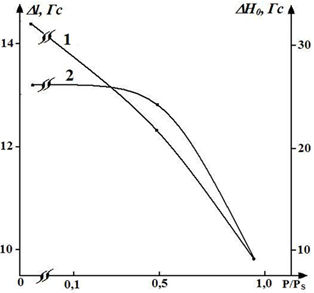
Fig. 3. Dependence of the parameters of the EPR spectra Δl (1) and ΔH0 (2) on the content of relative humidity P / Ps, samples of cotton fiber of grade A-2.
The intensification of mobility leads to averaging of inhomogeneities of the label environment and a decrease in the width of the spectrum line. Since 2A’z decreases simultaneously (Fig. 2, curve 2) and ∆H0 narrows (Fig. 3, curve 2), we can conclude that in this humidity range the radical motion is close to rotational. The position of the “transitional” moistened region is determined by the structure and degree of hydration of the polymers. In dry samples (P / Ps = 0.04), the “transitional” region is significantly biased toward the dry regions.
Thus, it can be concluded from experimental data that the spectral and dynamic parameters characterizing the mobility of the spin label depend on the degree of moisture of the cotton fiber samples. It was found that at low relative humidity (P / Ps = 0.04) these parameters reflect the monotonic amplification of low-amplitude high-frequency motions in the polymer. At a relative humidity of P / Ps ≥ 0.60, new components of the spectrum of labels appear relating to the rapidly rotating radical, which indicates the intensification of conformational movements and structural transitions in polymer chains.
Structural properties of cotton fiber grown from the selection variety “20-solodia istiyololyat”
The molecular labeling method was used to study the molecular structure of cotton fiber of the “20-malodium istiloliyat” breeding variety grown in Tajikistan. It was shown that a change in the EPR spectral parameters of the spin of labeled cotton fibers indicates the ordering of its molecular structure. It has been established that the amount of attached radical in the matrix of the fiber is larger compared to the amount of this radical in the matrix of other cotton fiber varieties studied previously. As is known, the spin label method is one of the most effective methods for studying the molecular structure of cotton fiber.
It was found that the mobility parameters of spin nitroxyl labels introduced into the fiber structure reflect the corresponding dynamics of the molecules at the point of their attachment. The conformational mobility of the macromolecular chains of fibrous cotton of the selection variety “20-content of Isstiloliyat” was studied. Samples were chemically modified by the hydroxyl groups of the fiber with a spin label according to the procedure. The first derivative of the EPR absorption signal V1 was recorded on an EPR-1306 spectrometer.
EPR spectra were recorded in standard molybdenum ampoules with an internal diameter of 3.0 mm, in which 25 mg of a spin-labeled sample was placed under the following conditions: microwave attenuation of 5 dB, magnetic field sweep amplitude of 200 Oe, magnetic field sweep speed of 40 E / min.
where is the distance between the external extrema of the EPR spectrum of the sample at a given experimental temperature; – the distance between the external extrema of the EPR spectrum of the sample at a temperature of 77K.
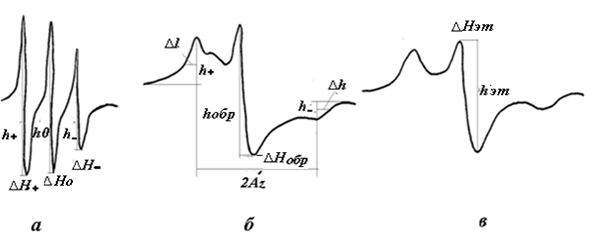
Fig. 1. EPR – spectra of nitroxyl radical (I); a- in an ethyl solution with a correlation time tc ≤ 10-7 s, b- spin-labeled cotton fiber of the “20-malaginium istololyat” cultivar with a correlation time tc ≥ 5 10-7s at room temperature and a reference 50% water- glycerin solution at 77K
Formula (1) is valid for the process of isotropic rotation of the nitroxyl fragment of the spin label by the Brownian diffusion mechanism. The actual rotation of the label cotton cellulose fragment in the cotton fiber macromolecule matrix can be anisotropic. The choice of a rotation model in the framework of the EPR spectroscopy data in the 3 cm range is a difficult, in many cases, unsolved problem. However, as shown in the literature, there is a direct proportional relationship between the value of tc calculated from theoretical spectra using a formula of the type (1) for isotropic motion and the values incorporated into the calculations for anisotropic rotation.
where Ret, Robr – the number of spin labels in the standard and the sample, respectively; het, hbr – respectively, the height of the central component of the EPR spectrum of the standard and sample; ∆Het, ∆Hbr — width of the central component of the EPR spectrum of the standard and sample in G. The calculation showed that the number of spin labels (R) attached to the polymer matrix of cotton fiber is 2.4 ∙ 1019 spin / g, that is, 2.4 ∙ 1019 spins correspond to one gram of the sample, which, compared to other varieties of cotton fibers, which we studied earlier by 1.5 times more. This indicates that the structure of the cotton fiber of the “20-malodium istiololiyat” variety is much better, more flexible and more informative.
According to the results of our work, it was found that the depth of the spin mark is about 13 Å for the varieties “Tashkent-1, Tashkent-2, 108-F, etc.”, and for the new variety “20-content of Istiololiyat” is 25 Å, which confirms the localization of the spin label in cotton fibers, the varieties of the “20-colodium of Istiololiyat to a greater depth.
2). The influence of environmental conditions on the molecular structure of medicinal plants of dandelion (Taraxacum officinale Wigg.) And chicory ordinary (Cichorium intybus L.
Brief information about the work performed.
The aim of this work was to study the influence of environmental conditions on the structure of molecules in various wild medicinal plants: dandelion (Taraxacum officinale Wigg.) And chicory ordinary (Cichorium intybus L.), growing in various regions of Tajikistan. A technique has been developed to study the chemical modification of molecules in wild-growing medicinal plants of dandelion and chicory using the spin label method. The stability and molecular mobility of the nitroxyl radical introduced into the matrix of dandelion and chicory were studied. It was found that at room temperature in the EPR spectra there is an inhibition of the rotational mobility of the spin label with a correlation time tc ≤ 10-7c, which indicates a change in the system of inter- and intramolecular hydrogen bonds in the region of attachment of the label. Changes in the parameters of the EPR spectra and rotational mobility of spin labels at room temperature depend on the environmental conditions of the plant growth site.
Medicinal plants are currently a valuable tool for the treatment and prevention of chronic diseases. Interest in medicinal plants is explained by the increased desire of people to be closer to nature, to avoid the negative effects of synthetic agents. At the same time, the advantage of most plants is their low toxicity. In addition, it is believed that medicinal plants that grow in environmentally friendly conditions are considered more effective [1]. Herbal raw materials are environmentally friendly and properly dried, rarely freshly picked medicinal plants (or parts thereof) used for the preparation of medicines [2]. It was shown in [3–11] that the growing conditions of medicinal plants can affect both the biosynthesis and the molecular state of the formation of the physicochemical structure of the substances that make up the plant, for example, the formation of a system of inter- and intramolecular interactions. In particular, it was concluded that the medicinal properties of plants are due to the accumulation of free radicals during biosynthesis, which largely depends on the environmental conditions of the place of growth. Therefore, it was important to study the molecular structures of the leaves and flower petals of plants living under conditions of constant exposure to a natural-technogenic radiation background, that is, the Pb-210 isotope, which is a decay product of natural uranium, and the technogenic Cs-137 isotope, by EPR spectroscopy. According to [5], these isotopes are accumulated in significant amounts in the gorge of the Siyoma River (a tributary of the Varzob River), about 50 km from Dushanbe (Fig. 1). In studying the structure of liquids, polymers, and biological structures, various physicochemical methods are used. However, many methods do not provide detailed information on the dynamic structure of molecules, changes in conformational transitions, and the content of various defects
and microvoids. In obtaining information on the dynamic structure of molecules, the spin label method based on stable nitroxyl radicals has found wide application [12–20]. Spin labels play the role of molecular sensors and provide unique information about the various dynamic changes in the system under study. In this work, we developed a method for studying the chemical modification of wild-growing medicinal plants of dandelion and chicory by a spin label, and the mobility of the spin label of modified studied objects at room temperature was also studied by EPR. Dandelion samples were collected during flowering in the places of growth: near and far from the motorway (Dushanbe), as well as at different heights above sea level and various conditions of constant exposure to natural and technogenic radiation background in the Siyom river basin (Varzob gorge, Republic of Tajikistan ), at the confluence with the tributary Small Igizak, Big Igizak and Igizak. Samples of chicory were collected in the Pamirs: Khorog, Darvaz, Shugnansky areas. Samples were thoroughly cleaned of the accompanying substances, washed with ordinary and distilled water, extracted with ether, alcohol and dried at room temperature.
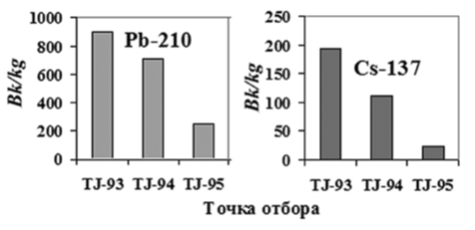
Fig. 1. The radiation dose distribution of the isotopes Pb-210 and Cs-137 at the sampling sites: TJ-93 (Small Igizak); TJ-94 (Big Igizak); TJ-95 (Igizak), (TJ-93-95 – reference designations of the place of gathering).
As a spin mark used a stable nitroxyl radical (I) having the following structural formula: